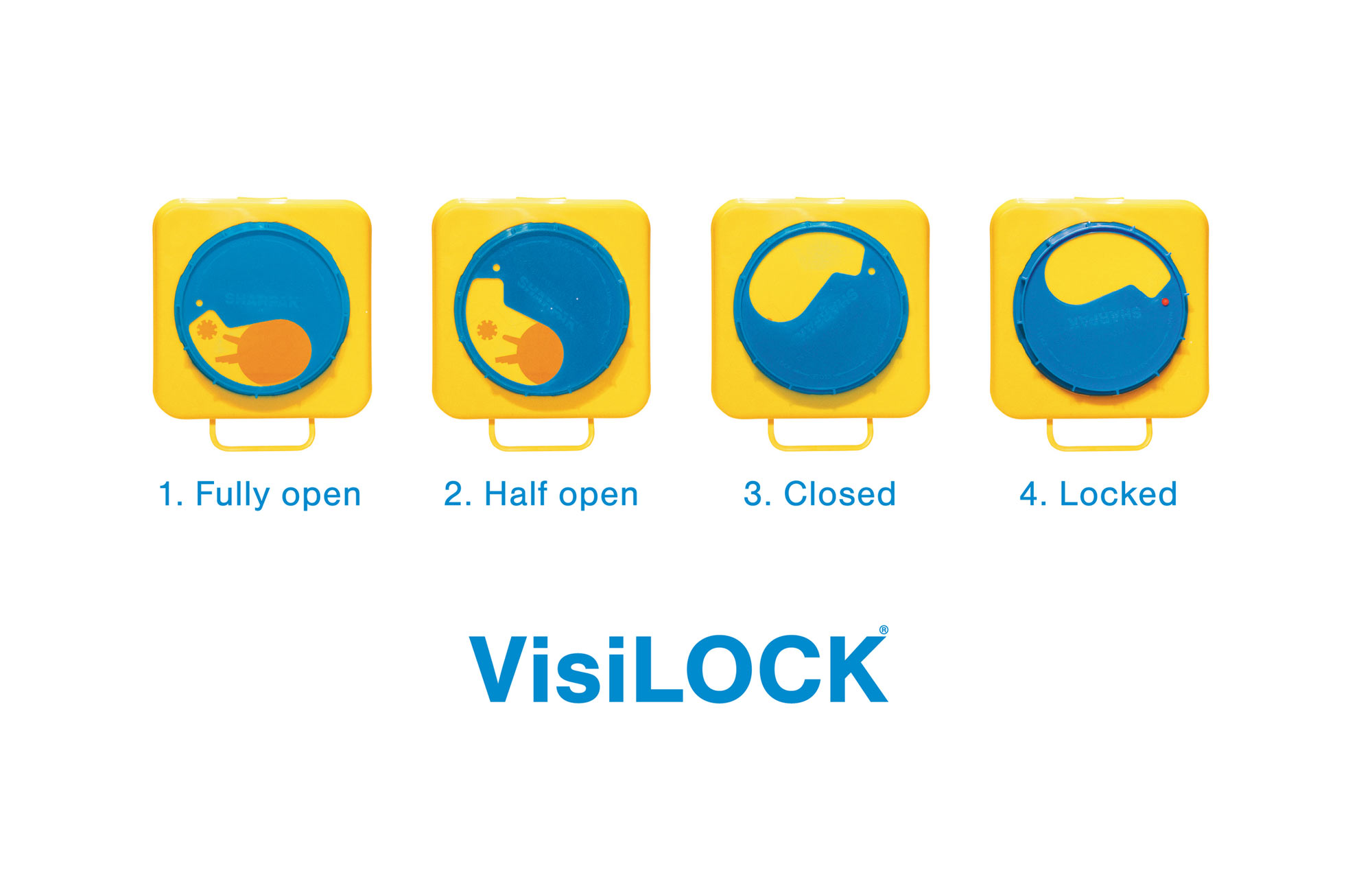
Sharpak® is the brand of clinical waste bins owned by Inpress, a leading medical packaging company and a wholly owned, family-run business based in Littlehampton on the south coast of the UK.
Our entire range is produced in our new 15,000 sq. feet centralised factory to maintain complete control and oversight. Our production runs 24/5 to ensure we meet our commitments to service our customers across the globe.